Filter by
News (79)
RSS
Applications are now open for the EDCTP Prizes 2027, recognising outstanding individuals and research teams from Africa and Europe for their achievements in global health research and innovation. Submissions are accepted until 2 September 2026.
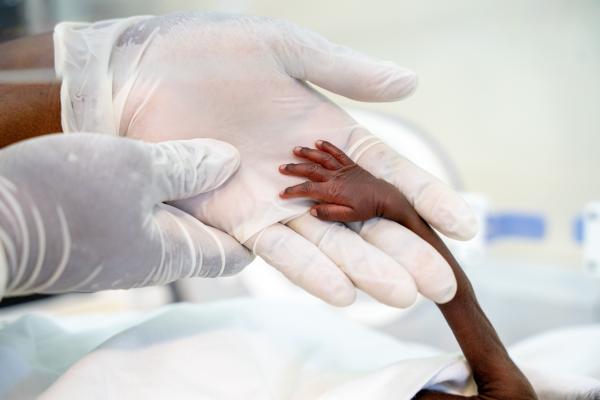
Coartem® Baby, co-funded by EDCTP2 through the PAMAfrica consortium, closes a long-standing treatment gap for one of the most vulnerable patient groups.
Global Health EDCTP3 supports 11 vaccine-focused projects worth €63 million to address persistent gaps in immunisation against infectious diseases.

The SAFIRE project, funded by Global Health EDCTP3, is generating first-of-its-kind clinical evidence to improve malaria treatment in early pregnancy.

Global Health EDCTP3 has received 277 applications by the 4 March 2026 first stage deadline for four topics of the 2026 calls. Successful consortia will be invited to submit a full proposal for the second stage by 17 September 2026.

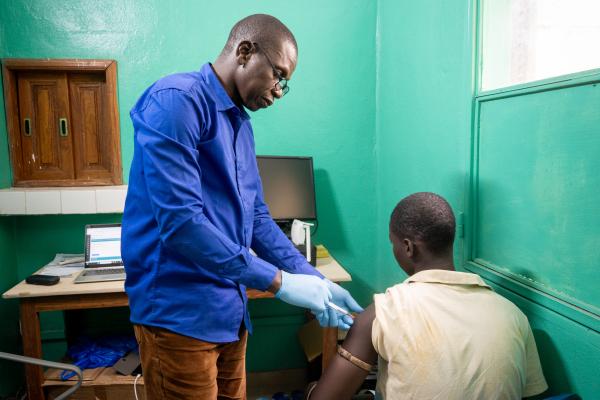
From malaria and HIV to pandemic preparedness and regulatory science, meet the EDCTP-funded scientists and leaders advancing research, strengthening health systems and building scientific capacity across Africa.

Results from the EDCTP2-funded Phase IIb trial demonstrate that the oral vaccine ETVAX significantly reduces moderate-to-severe diarrhoea in young children caused by E. coli.

Through collaboration with European health bodies, Global Health EDCTP3 aims to strengthen tuberculosis prevention and treatment, especially involving children and people with weakened immune systems.

The new Acoziborole Winthrop, supported by EDCTP, will help treat human African trypanosomiasis (HAT), commonly known as sleeping sickness.

The Global Health EDCTP3 Governing Board has appointed new members to its Stakeholders' Group, following an evaluation of 70 applications received from organisations worldwide. 32 African, European and international organisations have been appointed as members and three as observers.