In 2024, the estimated number of malaria cases climbed to 282 million, up from 273 million the previous year, with related deaths reaching approximately 610,000 according to the World Health Organization (WHO). While the world has seen historic successes such as the prevention of 2.3 billion cases and 14 million deaths since 2000 and the recent malaria-free certification of 47 countries and one territory, the disease remains a major burden for global health.
The WHO African Region continues to face the most acute crisis, accounting for 94% of all cases and 95% of deaths. This region's progress is increasingly jeopardised by additional threats such as the spread of antimalarial drug resistance, which is now confirmed or suspected in at least eight African countries, and the emergence of invasive mosquito species like Anopheles stephensi.
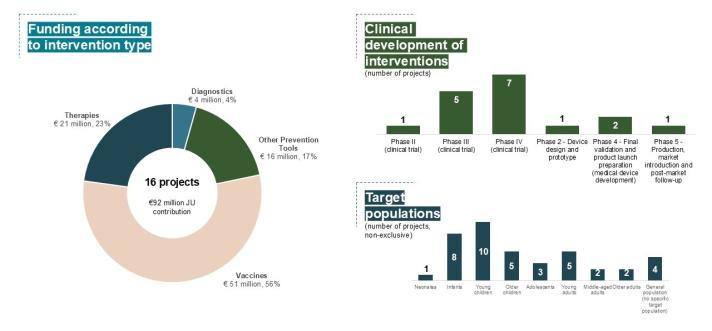
To date, Global Health EDCTP3 has invested €92 million across 16 projects on malaria research under the Horizon Europe programme. Our current malaria investment portfolio comprises €51 million allocated to malaria vaccines, €21 million on therapeutics, €4 million on diagnostics, and €16 million on other essential prevention tools.
'Global Health EDCTP3 supports Africa-Europe research teams that generate much-needed scientific evidence, including in populations often overlooked in clinical trials, such as pregnant women. Targeted research in high-burden regions saves lives and enhances health security, solidifying the EU's role in global health. We aim to accelerate R&D and improve access to medical interventions to drive progress toward malaria elimination.'
Michael Makanga, Executive Director, Global Health EDCTP3.
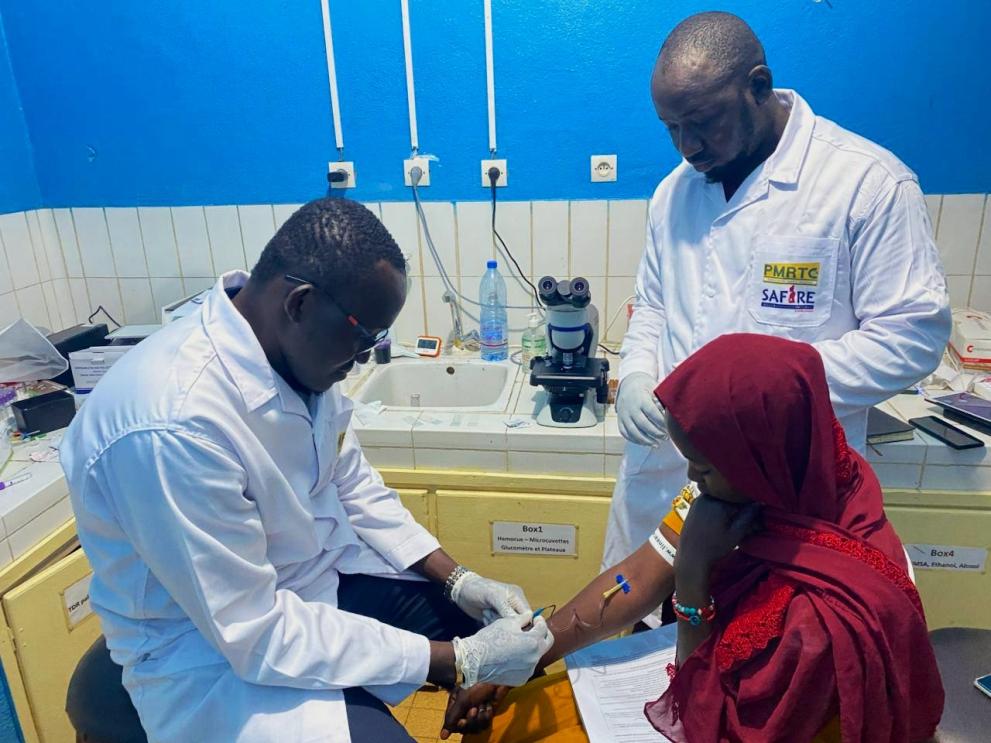
SAFIRE: The first-of-its-kind adaptive clinical trial
A clear example of this commitment is the SAFIRE project, Safety of Antimalarials in FIRst trimEster, an innovative initiative funded by Global Health EDCTP3 that is addressing one of the most significant ethical and evidence gaps in global health.
Every year, over 12 million pregnant women in sub-Saharan Africa are exposed to malaria, resulting in roughly half a million infections in the first trimester alone. Despite this significant burden, women in early pregnancy have traditionally been excluded from clinical trials due to safety concerns, leaving them with limited therapeutic options and depriving them of newer, more effective treatments.
For decades, the global health community relied on observational data that often lagged behind the needs of this vulnerable population. Even today, only one artemisinin-based combination therapy (ACT), artemether-lumefantrine, is recommended for use in the first trimester.
SAFIRE is conducting the first-ever phase III adaptive platform trial specifically designed for women in their first trimester. This innovative clinical trial design allows researchers to evaluate multiple treatments simultaneously, comparing the efficacy and safety of two commonly used ACTs, pyronaridine-artesunate and dihydroartemisinin-piperaquine, against the current standard of care.
The project reached a pivotal milestone in October 2025 with the enrollment of the very first patient in Mali, signifying the official start of this landmark study. Recruitment has since gained steady momentum, with currently 13 women enrolled in Mali, 31 in Burkina Faso, and 8 in Kenya.
'This first-of-its-kind trial addresses an important need for the treatment of malaria during the first trimester of pregnancy, being able to treat the infection with safe and effective drugs. SAFIRE project will provide an evidence base to make additional drugs available for malaria treatment in the first trimester, and also contribute to addressing the lack of equity in enrolling pregnant women in clinical trials.'
Dr Henk Schallig, SAFIRE coordinator, Amsterdam UMC.
These clinical achievements are the result of extensive preparation and community integration. The consortium, led by the Amsterdam University Medical Center, with scientific leadership from the University of Sciences, Techniques and Technologies of Bamako (USTTB) and the Kenya Medical Research Institute (KEMRI), has already secured all necessary institutional and ethical approvals across the implementing countries and started training local study staff in advanced laboratory procedures.
By generating high-quality safety data, SAFIRE aims to inform national and international treatment guidelines and equip healthcare providers with stronger tools to prevent maternal anaemia, stillbirth, and neonatal mortality, while also reducing the burden of malaria in pregnant women through evidence-based, equitable care.
Details
- Publication date
- 23 April 2026
- Author
- Global Health EDCTP3 Joint Undertaking